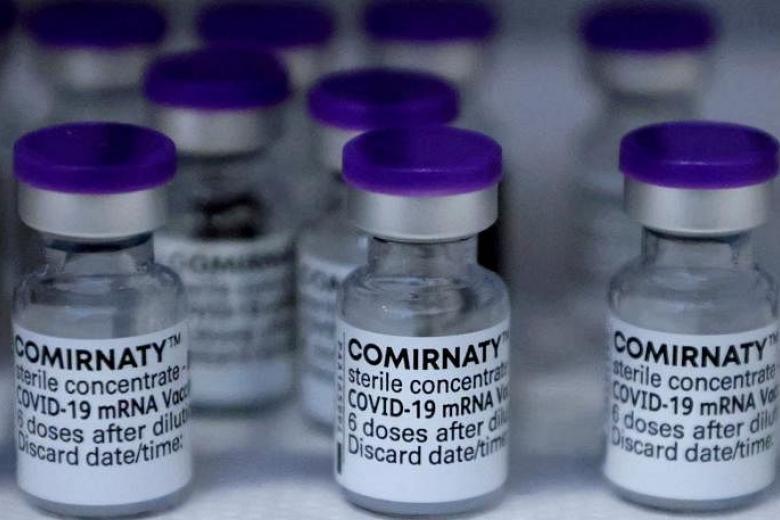
Pfizer Covid-19 vaccine approved for children aged 6 months to 4 years
In a statement, the Health Sciences Authority (HSA) said it had carefully considered the clinical data and assessed that the benefits outweighed the risks in administering the Comirnaty vaccine in children in this age group.
The vaccination regimen for the primary series in this age group will consist of three 3-microgram doses - the first two to be administered three weeks apart, followed by a third dose to be administered at least eight weeks after the second dose, said the HSA.
But it added: "Official vaccination recommendations on the use of this vaccine will be issued by the Expert Committee on Covid-19 Vaccination and the Ministry of Health when ready."
The authority also said that it consulted experts from its Medicines Advisory Committee and Panel of Infectious Diseases Experts in making the decision.
This is the second vaccine authorised for use in Singapore by HSA for children aged six months to four years old, after the Spikevax vaccine from Moderna.
The Spikevax vaccine was authorised for use on children aged six months to 17 years old on Aug 24.
As for the Pfizer vaccine, the authority said clinical data it considered included an ongoing Phase 2/3 study conducted by Pfizer, involving about 1,800 participants aged 6 months through 4 years. Read More…