Uranium trichloride exhibits transient covalency when hot
Uranium trichloride (UCl₃) is an intriguing compound known for its complex behavior under various conditions. This compound, a chloride of uranium, is typically used in nuclear chemistry and has properties that make it of particular interest to scientists studying actinides. One of the most fascinating aspects of uranium trichloride is its ability to exhibit transient covalency when heated. This phenomenon is not only scientifically significant but also essential for understanding the broader chemical properties of actinides and their potential applications.
Understanding the Structure of Uranium Trichloride
Before diving into the transient covalency exhibited by uranium trichloride, it's essential to understand its basic structure. UCl₃ consists of uranium in a +3 oxidation state, bonded to three chlorine atoms. The bonding in UCl₃ is predominantly ionic, with uranium and chlorine ions forming a lattice structure typical of many metal halides.
However, the behavior of this compound changes under specific conditions, such as elevated temperatures, where the electronic structure of uranium can lead to unique bonding characteristics.
Covalency in Uranium Compounds: An Overview
Covalency refers to the sharing of electron pairs between atoms, a type of chemical bonding typically seen in molecules like water (H₂O) or carbon dioxide (CO₂). In contrast, ionic bonds involve the transfer of electrons from one atom to another, resulting in the formation of ions. Uranium compounds, especially those with higher oxidation states, can sometimes exhibit covalent character due to the involvement of 5f, 6d, and 7s orbitals in bonding.
While uranium trichloride is primarily ionic at room temperature, it can exhibit covalent characteristics under specific conditions, such as when heated. This change is what scientists refer to as transient covalency.
What Is Transient Covalency?
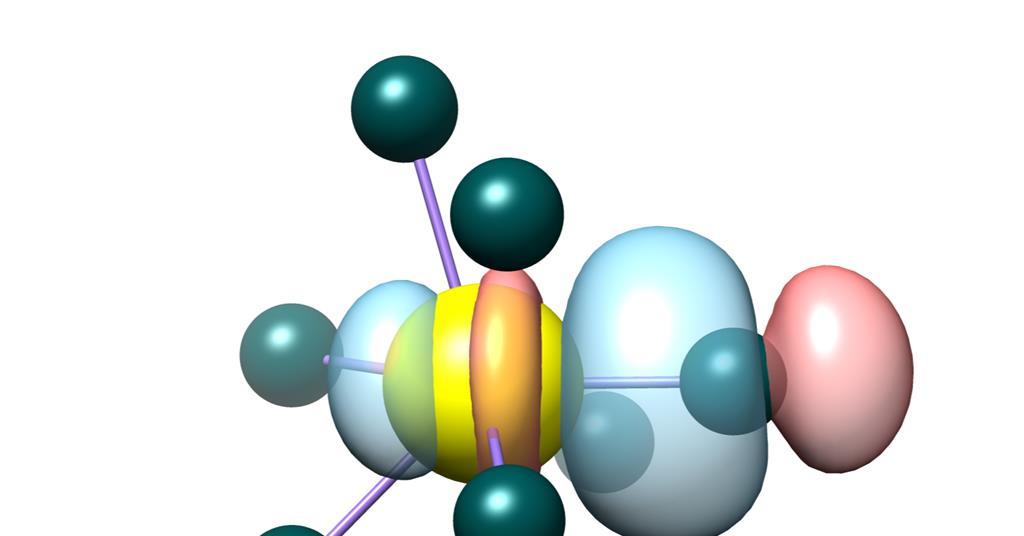
Transient covalency occurs when a compound temporarily exhibits covalent bonding characteristics under certain conditions, such as temperature changes, pressure, or the presence of a reactive species. In the case of uranium trichloride, transient covalency is observed when the compound is heated. At higher temperatures, the electrons in uranium's orbitals can become more delocalized, leading to a temporary sharing of electrons between uranium and chlorine atoms, thus exhibiting covalent bonding.
This phenomenon is transient because it only persists under the conditions that promote it, such as elevated temperatures. Once the compound cools, the electrons revert to their more localized states, and the bonding becomes predominantly ionic again.
Why Does Uranium Trichloride Exhibit Transient Covalency When Hot?
The transient covalency in uranium trichloride at elevated temperatures can be attributed to several factors:
- Electronic Structure of Uranium: Uranium's electronic configuration allows for the involvement of its 5f, 6d, and 7s orbitals in bonding. At higher temperatures, these orbitals can overlap more effectively with the orbitals of chlorine, leading to partial covalency.
- Thermal Activation: Heat provides the energy necessary to overcome the potential energy barriers between ionic and covalent bonding. This thermal activation allows for a temporary redistribution of electron density, resulting in covalent characteristics.
- Bond Length Changes: At higher temperatures, the bond lengths within the UCl₃ lattice can change, potentially allowing for better orbital overlap and hence covalent character.
Implications of Transient Covalency in Uranium Trichloride
The transient covalency exhibited by uranium trichloride when hot has significant implications for both theoretical and practical applications:
- Nuclear Chemistry: Understanding the bonding behavior of uranium compounds is crucial in nuclear chemistry, especially in the context of nuclear fuel processing and waste management. The transient covalency might affect the reactivity and stability of uranium compounds under operational conditions in reactors.
- Material Science: The covalent characteristics observed at high temperatures can influence the physical properties of materials containing uranium trichloride, such as melting points, electrical conductivity, and thermal stability.
- Actinide Chemistry: This phenomenon contributes to the broader understanding of actinide chemistry, highlighting the complexities and unique behaviors of these heavy elements.
Experimental studies have provided evidence of transient covalency in uranium trichloride through techniques such as X-ray diffraction, spectroscopy, and computational modeling. These studies typically observe changes in bond lengths, electron density distribution, and vibrational modes that indicate a shift towards covalent character at elevated temperatures.
Theoretical models, including quantum chemical calculations and molecular dynamics simulations, have been employed to explain the transient covalency in uranium trichloride. These models suggest that the covalent character arises from the interaction of uranium's 5f and 6d orbitals with the chlorine orbitals, facilitated by thermal energy.
It's essential to compare the behavior of uranium trichloride with other uranium halides, such as uranium hexafluoride (UF₆) and uranium tetrachloride (UCl₄). These comparisons help to highlight the unique bonding characteristics of UCl₃ and how they differ from compounds with different oxidation states or halides.
Studying transient covalency in uranium trichloride poses several challenges, primarily due to the need for high-temperature conditions and the radioactive nature of uranium. Experimental setups must be carefully designed to ensure accurate measurements while maintaining safety. Despite the challenges, the transient covalency of uranium trichloride has potential applications, particularly in nuclear technology and materials science. Understanding this behavior could lead to innovations in the design of new materials or the improvement of nuclear processes.
Uranium trichloride exhibits a fascinating chemical behavior known as transient covalency when heated. This temporary shift from ionic to covalent bonding provides valuable insights into the electronic structure and reactivity of uranium compounds. As research continues, the understanding of transient covalency in uranium trichloride could lead to significant advancements in various scientific fields, particularly in nuclear chemistry and material science.